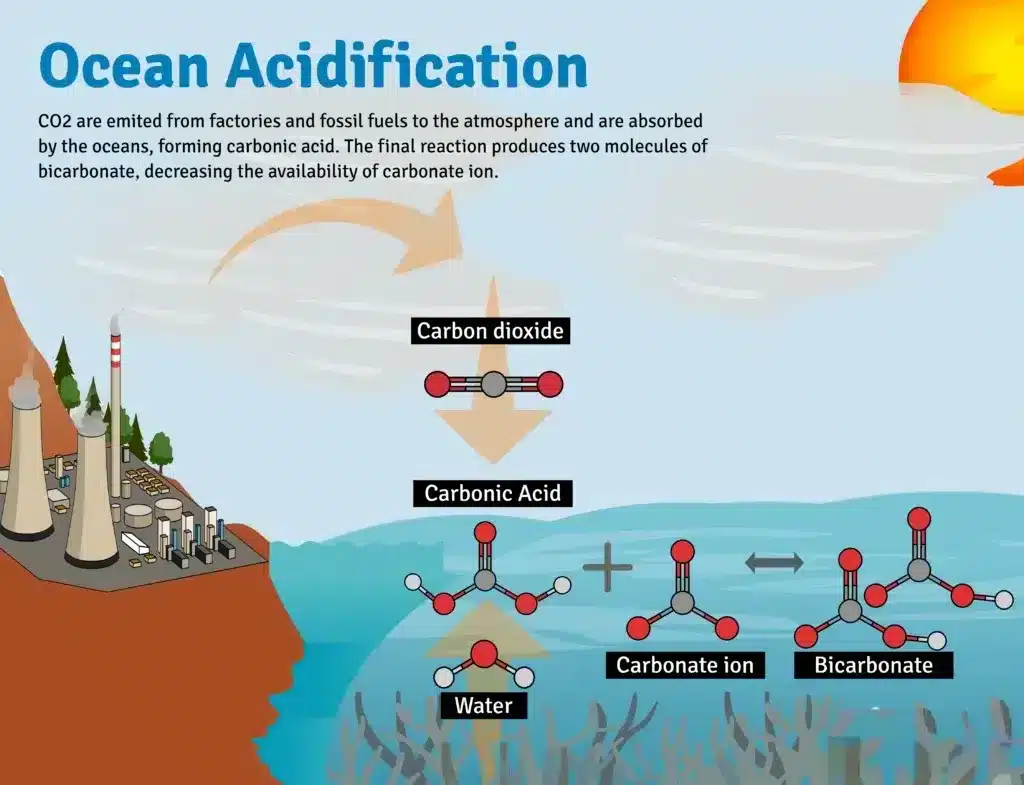
Ocean acidification refers to the ongoing decrease in the pH of the oceans, caused by the uptake of carbon dioxide (CO2) from the atmosphere. As human activities like burning fossil fuels release more CO2, the oceans absorb about 30% of it, increasing acidity. Since the Industrial Revolution, the pH of ocean surface waters has declined by 0.1 pH units.
Ocean acidification can have far-reaching impacts on marine ecosystems; on both shell-forming animals like corals, crabs, oysters, urchins, etc. as well as non-calcifying organisms. Tackling ocean acidification requires reducing CO2 emissions and protecting marine ecosystems.
About Ocean Acidification
Ocean acidification refers to the gradual decrease in ocean pH due to the absorption of carbon dioxide (CO₂) from the Atmosphere, primarily driven by fossil fuel emissions. Since pre-industrial times, atmospheric CO₂ levels have risen from 280 ppm to nearly 400 ppm, causing the ocean to absorb about 25% of these emissions.
- This process alters seawater chemistry, impacting marine ecosystems and biodiversity.
- While the ocean has mitigated global warming by storing excess CO₂, this buffering capacity is reaching its limits, posing serious environmental and socioeconomic risks.
Ocean Acidification Causes
The primary cause of ocean acidification is the increased carbon dioxide emissions from human activities, especially burning fossil fuels like coal and oil. Some key causes of ocean acidification are:
- Natural processes: Organic material, including the remains of sea organisms, accumulates on seabeds. When decomposed, these materials release CO₂, contributing to acidification.
- Chemical reactions at the seabed can also elevate hydrogen ion concentrations, further lowering pH levels.
- Chemical process: When CO₂ dissolves in seawater, it undergoes a series of reactions, forming carbonic acid (H₂CO₃). This weak acid dissociates into hydrogen ions (H⁺) and bicarbonate ions (HCO₃⁻).
- The increase in H⁺ ions results in more acidic water and a decline in carbonate ions (CO₃²⁻), which are essential for marine calcifiers.
- CO₂ absorption by the oceans: Oceans absorb approximately 30% of atmospheric CO₂, helping mitigate global warming but at the cost of increasing oceanic CO₂ levels and triggering acidification.
- Ocean water is mildly basic, with a pH of 8.0 to 8.2 pH units.
- Rising atmospheric CO₂: Human activities such as the burning of fossil fuels, deforestation, and industrialisation have significantly increased CO₂ levels.
- According to the Global Carbon Budget 2024, annual CO₂ emissions rose from 11 billion tons in the 1960s to 41.6 billion tons in 2024.
- Waste disposal: Industrial and domestic waste dumping in oceans lowers pH levels.
- Eutrophication: Caused by nutrient-rich runoff, it triggers massive plankton blooms. When these blooms collapse, bacteria decompose the organic matter, consuming oxygen and releasing CO₂ into the water.
- This process further lowers seawater oxygen levels and reduces the pH, aggravating acidification.
Ocean Acidification Influencing Factors
Ocean acidification, driven by rising atmospheric CO₂ levels, severely affects marine ecosystems. Various environmental factors influence this phenomenon such as:
- Sea Surface Temperature: A temperature rise accelerates ocean acidification due to increased CO₂ absorption in warmer waters.
- Salinity: Greater salinity reduces the solubility of CO₂, mitigating acidification to some extent.
- Particulate Organic Carbon (POC): Higher POC levels are beneficial, as organic carbon binds with CO₂, lowering free CO₂ concentration in water and mitigating acidification effects.
- Particulate Inorganic Carbon (PIC): High PIC concentrations aggravate acidification by increasing carbonates, which release CO₂ upon dissolution.
- Dissolved Oxygen: A decline in oxygen levels may aggravate acidification by destabilising carbonate compounds essential for marine organisms.
Ocean Acidification Impacts
Ocean acidification, caused by increased CO₂ absorption, has reduced ocean pH from 8.2 in pre-industrial times to 8.1 today, with projections indicating a further drop of 0.3–0.4 units by 2100. This seemingly minor change reflects a 30% rise in acidity, disrupting marine ecosystems and vital chemical processes.
- Oceans as carbon reservoirs: Oceans absorb about one-third of CO₂ emissions from human activities, playing a vital role in climate buffering. However, the rate of CO₂ uptake now exceeds the natural buffering capacity of marine ecosystems.
- Sulphur cycle and clouds: Oceans are the primary source of atmospheric sulphur, mainly in the form of dimethylsulphide (DMS) produced by phytoplankton.
- DMS enters the atmosphere, forming sulphuric acid that aggregates into aerosols, which act as cloud condensation nuclei.
- Clouds, in turn, reflect sunlight, playing a cooling role in Earth's climate system.
- Acidified oceans reduce DMS production by phytoplankton, potentially decreasing cloud formation and increasing global temperatures due to reduced solar reflection.
- Biodiversity and Marine Life:
- Shell builders and calcifiers: Organisms like oysters, clams, sea urchins, corals, and plankton rely on carbonate ions to form their shells and skeletons. Reduced availability of these ions makes their survival and growth challenging.
- Fish and Seaweed: Acidic waters impair some fish species’ abilities, such as clownfish, to detect predators or locate habitats. In contrast, algae and seagrasses may thrive due to higher CO₂ levels, benefiting from enhanced photosynthesis.
- Planktonic ecosystems: Phytoplankton, which form the base of marine food webs, may face disruptions, affecting the entire ecosystem.
- Environmental and Economic Impacts:
- Food web disruption: Acidification causes the extinction or decline of key species, affecting dependent organisms and disrupting marine food webs.
- Food shortages: The death of marine species threatens the livelihoods of communities dependent on fisheries. By 2100, molluscus losses could cost over $100 billion annually.
- Tourism decline: Damage to ecosystems like coral reefs, such as Australia's Great Barrier Reef, can significantly impact tourism revenue, estimated at A$5.4 billion annually.
Ocean Acidification in the Indian Ocean
Ocean acidification is increasingly evident in the Indian Ocean, a region vital for global biodiversity and the livelihoods of millions. The unique geographical, climatic, and ecological characteristics of the Indian Ocean make it particularly vulnerable.
- Arabian Sea: High CO₂ absorption aggravates acidification, impacting one of the most fertile marine regions. Pollutants from the Indo-Gangetic plains mix with seawater, further lowering pH.
- Bay of Bengal: Pollutant deposition during winter contributes to increasing acidity. Studies show a decline in phytoplankton, a critical base for the marine food chain.
- Regional risks: The Indian Ocean could become an ecological desert due to acidification and warming trends.
Initiatives to Curb Ocean Acidification
Tackling ocean acidification is vital to achieving the UN Sustainable Development Goal (SDG) 14, which emphasises the sustainable use of oceans and marine resources. Target SDG 14.3 specifically focuses on reducing the impacts of acidification through enhanced scientific collaboration and coordinated global action. Key initiatives include:
- Rio+20 Summit (2012) emphasised ocean acidification as a critical threat and highlighted oceans in sustainable development.
- Intergovernmental Oceanographic Commission (IOC) of UNESCO supports global coordination to minimize acidification through efforts like the Global Ocean Acidification Observing Network (GOA-ON).
- International Atomic Energy Agency (IAEA) established the Ocean Acidification International Coordination Centre (OA-ICC) to advance science and policy solutions.
- High Seas Treaty: It covers 64% of the ocean and faces overexploitation and biodiversity loss due to being a global commons.
- A proposed treaty, likened to the 2015 Paris Agreement, aims to protect vast marine areas and ensure the sustainable use of ocean resources.
- Scientific and local efforts: Enhancing Marine Protected Areas (MPAs) to safeguard critical ecosystems. Examples include MPAs in Antarctica and proposals to expand protection in regions like the Weddell Sea. Promoting sustainable fishing practices to ensure marine ecosystem stability.
Ocean Acidification Way Forward
A proactive and multi-dimensional approach is required to mitigate ocean acidification impacts. The following are key strategies for a sustainable way forward:
- Reducing CO₂ emissions: Shift to renewable energy sources like solar and wind to minimize reliance on fossil fuels. Encourage the adoption of carbon-neutral technologies and energy diversification.
- Carbon Capture and Storage (CCS): Invest in technologies to capture CO₂ emissions from industrial processes and power plants before they enter the atmosphere
- Raising awareness: Educate the public on the significance of ocean acidification and its cascading effects on ecosystems, food security, and livelihoods.
- Sustainable development: Invest in scientific research to better understand acidification trends and their implications. Promote international collaboration through initiatives like Blue Carbon Initiatives, GOA-ON and IOC of UNESCO.
Ocean Acidification PYQs
Question 1: The acidification of oceans is increasing. Why is this phenomenon a cause of concern? (UPSC Prelims 2012)
- The growth and survival of calcareous phytoplankton will be adversely affected.
- The growth and survival of coral reefs will be adversely affected.
- The survival of some animals that have phytoplanktonic larvae will be adversely affected.
- The cloud seeding and formation of clouds will be adversely affected.
Which of the statements given above is/are correct?
a) 1, 2 and 3 only
b) 2 only
c) 1 and 3 only
d) 1, 2, 3 and 4
Answer: (d)
Last updated on March, 2026
→ UPSC Final Result 2025 is now out.
→ UPSC has released UPSC Toppers List 2025 with the Civil Services final result on its official website.
→ Anuj Agnihotri secured AIR 1 in the UPSC Civil Services Examination 2025.
→ UPSC Marksheet 2025 is now out.
→ UPSC Notification 2026 & UPSC IFoS Notification 2026 is now out on the official website at upsconline.nic.in.
→ UPSC Calendar 2026 has been released.
→ Check out the latest UPSC Syllabus 2026 here.
→ UPSC Prelims 2026 will be conducted on 24th May, 2026 & UPSC Mains 2026 will be conducted on 21st August 2026.
→ The UPSC Selection Process is of 3 stages-Prelims, Mains and Interview.
→ Prepare effectively with Vajiram & Ravi’s UPSC Prelims Test Series 2026 featuring full-length mock tests, detailed solutions, and performance analysis.
→ Enroll in Vajiram & Ravi’s UPSC Mains Test Series 2026 for structured answer writing practice, expert evaluation, and exam-oriented feedback.
→ Join Vajiram & Ravi’s Best UPSC Mentorship Program for personalized guidance, strategy planning, and one-to-one support from experienced mentors.
→ Shakti Dubey secures AIR 1 in UPSC CSE Exam 2024.
→ Also check Best UPSC Coaching in India
Ocean Acidification FAQs
Q1. What do you mean by ocean acidification?+
Q2. What are the effects of ocean acidification?+
Q3. What is the pH of ocean acidification?+
Q4. Does ocean acidification affect cloud seeding?+
Q5. Does the greenhouse effect cause ocean acidification?+
Tags: quest UPSC Geography Notes